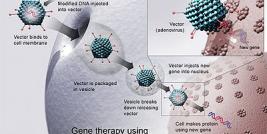
The year 2009 was quite turbulent for gene therapy companies, with lay-offs, bankruptcies and general financial crisis. Earlier 2008 FDA refused US based Introgen’s Biologics License application (1) and during 2009 now bankrupt Introgen sold it’s GMP facilities and other remaining assets to Vivante GMP Solutions, ceasing to exist as gene therapy company. Similarly other gene therapy companies have had difficulties on getting their products to the market, especially with the economic crisis hindering their financing. After facing extensive difficulties, the path often leads to turning into a CMO, field with heavy competition.
There are still some promising signs of new interest rising towards gene and cell therapy. Big Pharma seems to be more active to open discussions with innovative new gene therapy companies and progress to the marketing approval-stage by several companies has convinced the investors that soon the first product should be on the markets also in the Western World.
Targeted Genetics
One of the oldest gene therapy companies, Targeted Genetics faced near-bankruptcy and was dimished to manufacturing core facility during 2009. With a deal with Genzyme, the company sold it’s intellectual properties and acquired enough cash to sustain it’s activities through 2010. With some promising products to various diseases, there is still hope that they would some day end to markets via partner (2).
Ark Therapeutics
Another company with gene therapy products, Anglo-Finnish Ark Therapeutics, filed to a marketing authorization approval for it’s lead product Cerepro® November 2008. The company received negative results from EMA December 16th 2009, suffering a huge blow to its stock price and future plans for gene therapy.
EMA’s opininion raised a question about the impartiality of one of the sites, stating that there were suspicions that patients receiving Cerepro®, were waiting for the re-operation longer than control group, thus giving Cerepro® more time to function. In addition, there were also concerns of side-effects, including slight paralysis and seizures. (3)
After receiving the negative opinion, company’s stock price diminished from 37 pence to 12 pence, gaining a few pences when the company announced applying for an appeal to EMA. The results for this appeal are expected at Q2 of 2010. In addition, very shortly after the negative opinion, the Finnish subsidiary of Ark Therapeutics announced lay-offs concerning 50 % of it’s staff in Finland, likely in the RD and production support staff (4).
The company had previously reported signing contract manufacturing deals with two clients for manufacturing adenovirus-based drugs. (5) Given the fact that company built a custom-made production facilities to Finland to produce Cerepro® and a another pipeline-product TrinamTM, this would perhaps indicate that company was already preparing for a dimished production. With surprising plans of 50 % personnel reduction before the appeal results are known, it seems that the company has very little faith passing it’s appeal to EMA.
While the company has income from it’s wound care-unit, plans for it’s second product-in-pipeline TrinamTM, VEGF-D containing adenoviral drug, would suffer severely, possibly forcing the company to seek partners for development. These negotiations are propably not made any easier by Circadian Technologies, which announced late October 2009 that they are starting legal actions against Lymphatix oyj, previously acquired by Ark Therapeutics and terminate the VEGF-D licence for both Ark Therapeutics and Lymphatix. Ark considers that it does not require the Ark License as it owns Lymphatix, which it considers has the rights to market TrinamTM using VEGF-D intellectual property (6). During Q2 of 2010, Ark Therapeutics will receive opinion to it’s appeal and the future for it’s main products will be revealed. Hopefully the hard efforts for bringing the product to markets will be rewarded.
Amsterdam Molecular Therapeutics – AMT
Following Ark’s lead, a Dutch gene therapy company AMT, established 1998, filed it’s main product Glybera® to EMA late 2009. Glybera® is a AAV-based gene therapy product against a rare disease called lipoprotein lipase deficiency, LPLD.
In addition, AMT has a product pipeline with several AAV-based gene therapy products in Hemophilia B, DMD, Acute Intermittent Porphyria and Parkinson's Disease at different stages of research or development. The company is expecting the EMA opinion early 2011.
Interestingly, the company revealed 2009 that it had signed development and commercialization agreement for LPLchipTM, a diagnostic tool to rapidly diagnose patients with complete and partial lipoprotein lipase deficiency with Spanish company Progenika SA (7). Since it’s main focus is on a chronic disease, which is rare and had prevalence of 1-2: 1 000 000 (8), an rapid and affordable diagnostic test seems essential on patient identification. It is likely that in the future smaller gene therapy companies will have to create innovative business models and supportive product portfolios to secure steady cash-flow during the long development time.
Gene therapy product already in the markets
Due to the various economical and political reasons, China was the first country moving on with the gene therapy commercialization(9). Both of the currently available products (OncorineTM and GendicineTM) have their Western counterparts, but the acceptance process and results from the treated patients have raised concerns over different scientific and regulatory standards during the process (10). Interestingly, currently SiBiono Genetechnologies is owned by US listed Benda Pharmaceuticals and Sunway Biotech is owned by US Mergen Biotech. It remains to be seen whether these product will be expanded to Western markets at some point.
Gendicine
Gendicine™, a p53 adenovirus for the treatment of head- and neck squamous cell cancer is used in combination with radiotherapy (11,12). After the approval of Gendicine™ by the SFDA, there has been intensive discussion about the efficiency of the treatment and lack of details in the reporting (13,14,10).
This replication-incompetent adenovirus contains the p53 transgene in place of the viral E1 region, under a Rous sarcoma virus promoter together with a bovine growth hormone poly(A)-tail, produced in a bioreactor using human embryonic kidney cells and purified with chromatographical methods.
In a phase I clinical trial with 12 laryngeal cancer patients, only one patient resulted in self-limited fever and none of the patients had tumor relapse during the 5 year follow-up after the treatment (15,10). Similarly in phase II/III trials with 132 head and neck squamous cell carcinoma patients, 32% showed fever as the only side-effect of the treatment. When Gendicine™ was used in combination with radiotherapy, 64 % of the patients responded with a complete regression and 29 % with a partial regression while with radiotherapy alone, 19 % showed a complete regression and 60 % a partial regression, suggesting synergistic effect of the combination treatment (16). The regression of patients was evaluated by tumor shrink rates according to WHO response criteria. However, the published details of these clinical trials are limited, making comparisons to other cancer trials difficult (16,10,13).
Due to the various roles of p53 protein, the authors suggested that the results derive from the role of p53 in sequence-specific transcription regulation inducing apoptosis in nucleus, mitochondria and Golgi apparatus; activating the immune system, inhibiting DNA repair and downregulating drug resistance-genes and vascular endothelial growth factors (16).
The latest released data of Gendicine indicate that out of 26 patients with Gendicine and radiotherapy, 17 have survived for 5 years and 16 without any recurrence, while in the control group of 27 patients 14 survived for 5 years with 10 without any recurrence. These numbers are too small to draw any conclusions about the efficiency of Gendicine, yet there are claims that over 7000 patients have already been treated but there is no data about official phase IV study (10).
Oncorine
Oncorine™ for treating head and neck cancer (H101) is a conditionally replicative adenovirus, with a deletion in E1B 55K region (17,18,19), therefore restricting the virus to bind and inactivate wild-type p53 protein. Inactivation of the host cell p53 is essential for wild-type adenoviruses to disable the activation of apoptotic pathway when host cell shifts to S phase in the lytic infection. When E1B 55K activity is removed, the replication in normal cells is blocked, allowing only replication in p53-deficient cells (20). In malignant cells the viral proliferation leads to oncolysis, used as a cancer therapy to treat solid tumours.
In phase I trials, Oncorine™ showed tumor shrinking with 3 patients out of 15 with good safety (1 partial regression and 2 with minor response). With phase II trials of 53 patients, the treatment group (two cycles of intratumoral injection of Oncorine™) showed 28 % response rate against the 12 % of control (no data available), again well tolerated (3 with complete regression and 11 with partial regression). Finally, in phase III trials with 123 patients in treatment group (combination of chemotherapy and Oncorine™) or control (chemotherapy alone), the response rate in treatment group was 72.7 %, as compared to 40.4 % of the control group. Similarly with nasopharyngeal cancer, combination treatment with chemotherapy and Oncorine™ resulted in response rate of 75.6 % against 57.1 % of the control group. The response was evaluated by the shrinkage of the tumor according to the WHO criteria (18).
The patient’s complications included fever, injection site pain, nausea, alopecia, leucopenia and flu-like symptoms. The authors speculate that since patients with fever showed increased response rate, the elevated temperature might improve viral replication through heat shock protein assisted late mRNA export (18). The criticism towards the phase III trials is focused on the small number of patients in the trial (10).
Cerepro
Cerepro® (Sitimagene ceradenovec) from Anglo-Finnish company Ark Therapeutics Group PLC is an adenovirus containing a Herpes simplex type-1 thymidine kinase transgene, under the cytomegalovirus promoter, for the treatment of malignant glioma together with ganciclovir (21). The company claimed an Orphan Drug status for Cerepro® from EMA and FDA.
In phase I clinical trials adenovirus transduction efficiency in glioma tumor was evaluated to be overall from 0.01 to 11.3 % (22), while a later animal experiments with rat glioma model suggested that transduction efficiency over 10 % was needed for tumor shrinkage (23). A later phase I/IIa trial compared the efficiency of PA317/tk retroviral packaging cells vector to Cerepro® or LacZ patient groups (7 patients each), resulting significantly longer survival time with Cerepro® (15 months) as compared to LacZ patient group or (8.3 months) retroviral group (7.4 months).
After Market Authorization Application (MAA) to EMA in 2005, with limited patient numbers, CHMP gave 2007 a somewhat expected negative opinion with concerns of the low number of patients in the study and insufficient safety data, causing withdrawal of the application and eliminating the hopes for early approval of Cerepro® (1). In details, CHMP expressed concern over methodical flaws, heterogeneity of population as to prognosis and small sample size., causing the company to start phase III clinical trials in 2005 with 236 patients in 40 centers in EU and Israel.
In the phase III study the patients were randomised to either standard care plus Cerepro®(R) or standard care alone. Standard care was surgery and radiotherapy or surgery and radiotherapy followed by temozolomide, depending on the investigating centres' standard practice and patient suitability, giving four treatment groups. It showed a 42 day improvement in median survival (310 days vs 268 days) and the improvement over standard care reached significance (p<0.032). On the primary endpoint, the group given Cerepro®(R) and temozolomide showed an improvement of 68% in median survival time compared with standard care surgery and radiotherapy controls (350 days vs 208 days). Against the same controls, treatment with Cerepro®(R) alone showed an improved median survival trend approaching 50%, similar to those given treatment with temozolomide alone after surgery and radiotherapy (300 days and 307 days respectively vs 208 days with standard care). At 2009 Cerepro® was given named patient permission in France and Finland by the national medicinal agencies, but in the centralized procedure EMA gave negative opinion against Cerepro®, once again expressing concern over study design and also safety of the treatment. The company is currently in an appeal process with EMA.
Glybera
Glybera® from AMT biopharma is an adeno-associated virus containing S447X mutated LPL transgene with CMV-WPRE promoter for lipoprotein lipase deficiency. LPLD is a seriously debilitating, and potentially lethal, orphan disease. The disease is caused by mutations in the LPL gene, resulting in highly decreased or absent activity of LPL protein in patients. Recurrent pancreatitis in LPLD patients can result in difficult-to-treat diabetes.
The clinical trials of Glybera® started at 2005 with a phase I study with 10 patients in Netherlands and continued to phase IIa in Canada and phase IIb, also in Canada. The clinical outcome of the phase I study was that during the number of episodes reduced from 0.49 (within 6.4 patient years) to 0.04 (within 23.2 patient years). Similarly, in phase II trials, the number of episodes was reduced from 0.27 (within 14.8 patient years) to 0.07 (within 15 patient years). Amsterdam Molecular Therapeutics filed marketing approval for Glybera® late 2009 to EMA.
Disclaimer: The author is former employee of Ark Therapeutics and current employee of AMT.