Article by: Pierre Cordelier
Rationale
Human Immunodeficiency Virus (HIV)-derived lentiviral vectors provide efficient gene transfer in proliferative and quiescent cells and demonstrate stable, high-level transgene expression both in vitro and in vivo. HIV non specifically integrates its DNA into the human genome, with a preference for active genes. However, integration can be problematic because a variation in gene expression between cells, possible gene silencing, and most importantly insertional mutagenesis, that can provoke malignant transformation. These findings clearly indicate that integration of lentiviral vectors must be prevented to ensure optimal safety.
What are lentiviral vectors?
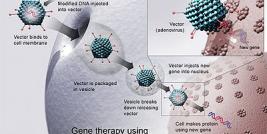
HIV-based lentiviral vectors are very promising for gene delivery because of their relatively large packaging capacity, reduced immunogenicity and their ability to stably transduce efficiently a large range of different cell types including quiescent cells. Lentiviral vectors (LV) are generated following transient transfection of three (packaging, envelope, and transfer) or more plasmids into producer cells. Packaging plasmids are deleted for many if not all viral coding sequences. As off, viral proteins required in trans are split in two or more plasmids, one encoding for gag/pol and the other one for rev. Transfer plasmids can also be modified to increase biosafety. Self-inactivating vectors (SIN) are generated following deletion of the promoter sequence (encompassing the viral enhancer and TATA box) in the 3’-long terminal repeat (LTR) U3 region. In these vectors, transgene expression depends entirely on internal promoters. To broaden the tropism of the vector, a third plasmid is used that encodes a heterologous envelope protein. Most of the time, lentiviral particles are pseudotyped with the fusogenic glycoprotein of vesicular stomatitis virus (VSV-G). The latter envelope offers the additional advantage of high stability, which allows for particle concentration by ultracentrifugation.
How lentiviral vectors do work
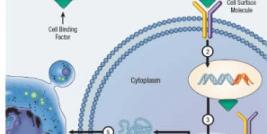
Like HIV, LV enter the target cell through the interaction of viral surface glycoproteins with receptors on the cell surface. On entry, the viral RNA undergoes reverse transcription, which is mediated by the viral reverse transcriptase complex. The product of reverse transcription is blunt-ended, double-stranded linear viral DNA, which is the substrate for viral integration in the DNA of infected cells. Despite their efficacy for gene transfer, nonspecific integration of retroviruses is a serious drawback to their use in clinical protocols because of the variation in gene expression between cells, possible gene silencing, and most importantly insertional mutagenesis, that can lead to undesirable effects such as malignant transformation. As a matter of fact, recent studies suggest that LV integration may triggers abnormal proliferation by activation of proto-oncogenes or disruption of anti-oncogenes either through a LTR or an internal promoter. In conclusion, for many application, preventing integration is the most straightforward approach to overcome the problem of malignant transformation.
Engineering non integrating lentiviral vectors: let it roll...
HIV-1 integration is driven by the ability of the lentiviral preintegration complex (PIC) to enter the cellular nucleus. This complex is composed of the linear viral DNA, the viral integrase (IN), reverse transcriptase (RT), matrix protein, the accessory protein vpr and miscellaneous cellular proteins. Lentiviral integration is mediated by the 32-kDa IN protein, which is encoded by the pol gene. HIV IN binds to chromosomal DNA and initiates integration. IN bounds to the attachment (att) regions of the LTRs to catalyze the covalent linkage of viral genome to the cell genome after DNA cleavage and joining activities . While this process is highly effective, nearly half of the nuclear proviral DNA becomes episomal: those circular forms of viral double-stranded DNA are produced during viral replication, together with free linear form and the integrated provirus. The two major circular episomial species result either from nonhomologous end-joining (NHEJ) and homologous recombination: they both encode for the whole viral genome but feature either one (1-LTR circle) or two (2-LTR circle) tandem LTR motifs. These viral episomes lack replication signals and are progressively diluted out. Altogether, there is exciting potential for effective use of non integrating LV (NILV) in gene transfer approaches to achieve therapeutic gene expression and significant safety benefits over their integrative counterparts.
Thus, viral IN is a very attractive target to make LV non integrative. However, IN cannot be deleted completely because it is also involved in the reverse transcription of HIV-1 RNA, nuclear import of the PIC, particle budding and viral DNA synthesis. Two class of IN mutation are described: a) class I mutations that specifically block integration, and b) class II mutations with pleiotropic effects on the virus replicative cycle. Clearly, class I mutations are preferred to impair the integration ability of LV without affecting levels of viral DNA. The catalytic domain of HIV-1 IN contains the three conserved amino acid D, D and E (D64, D116, E152). Class I IN-deficient NILV can be produced by introducing mutations in any of this amino acid triad, to specifically inhibits the integration of viral DNA into the host genome, without reducing DNA synthesis, nor disturbing gag-pol functions. Another option to prevent LV insertional mutagenesis is to mutate the att site in the viral genome. Mutating either U3 or U5 att sites reduced integration to 10-30% of wild-type levels only. Interestingly, combining both IN and att mutations do not result in further reduction of integration frequency. In summary, targeting IN results in NILVs residual integration frequencies 3 to 4 log below those of the wild-type, integration-prone LV. As a consequence, NILVs episomes copy number is greatly increased. Estimates of stable integration frequency of NILVs in cultured cells is equivalent to plasmid transfection (0.1 to 2.3%). Another concern is the possibility that NILVs can be susceptible to replication-competent retroviruses (RCR) and vector mobilization, i.e. complementation with wild-type HIV-1. However, IN-deficient NILVs are unlikely to support HIV-1 replication, a mechanism highly dependent on the IN activity. NILV mobilization can be more problematic. NILVs generate full length, integration and encapsidation-competent transcripts. One way to address this issue is to generate att mutants to reduce integration frequencies of NILVs mobilized by wild-type HIV-1. Taken together, catalytic active site IN mutations are the most efficient and clever approaches to generate NILVs.
NILVs drive efficient gene expression.
The design of NILV has long been hindered by the belief that episomal forms of LV were very unstable, did not transduced cells efficiently, nor supported transcription. It is now well established that NILV transduction efficiency is only slightly lower than integrative vectors. In our experience; NILV bearing D64V or D116N IN mutation transduce replicating cells with high efficacy (Cordelier et al, personal observation). As a matter of fact, early work reported poor transduction efficiency with non-SIN LV, later rescued by the outcome of SIN NILV. Following circularization by host enzymes, NILV are functional templates for transcription by host cell machinery. Concerning the strength of expression, recent reports demonstrate that transgene expression from NILV is very divergent and is 2 to 10 fold weaker than from integrated provirus in cell lines, but equivalent in vivo. Applications and ongoing development of NILVs include but are not restricted to transient gene expression, vaccination, RNA interference, homologous recombination, site-specific integration, transposition, generation of replication-specific NILVs… Surprisingly, their use as cancer gene therapy vehicles is still to be reported. Preliminary results obtained in our lab indicate that NILVs transduce cancer cells with high efficacy to ensure the transient expression of marker genes. We are in the process of designing “therapeutic” NILVs to ask whether they may drive successful in vivo cancer gene therapy.
Conclusions and future directions.
In conclusion, NILVs are very efficient gene delivery vectors, do not integrate frequently and, as a consequence, have much lower risk of insertional mutagenesis than their integrative counterparts. They represent a major advance to ensure safe and efficient gene transfer as compared to existing gene delivery vehicles. On the other hand, one major drawback of NILV may reside in limited transgene expression as compared to integrating LV. While they were designed to ensure stable and safer expression in non-dividing cells, it is tempting to speculate that these “super DNA minicircles” may be applied in a near future to the gene therapy of solid tumors.
Address correspondence to:
Dr Pierre Cordelier,Inserm U858, I2MR,CHU Rangueil,1, avenue Jean Poulhès.BP 84225, 31432 Toulouse Cedex 04, France.Tel: (33) 5 61 32 24 04 ; Fax: (33) 5 61 32 24 03.Email: pierre.cordelier@inserm.fr