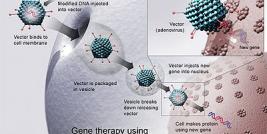
ALACHUA, FL May 16, 2012 - Florida Biologix®, a full-service biopharmaceutical development and contract manufacturing organization, provided cGMP manufacturing services to National Taiwan University Hospital under a contract signed in March 2008. Florida Biologix produced, purified, and tested a clinical batch of their novel gene therapy for a Phase I clinical trial of a rare genetic disease – AADC deficiency. The trial was led by Dr. Paul Wuh-Liang Hwu. Results of the clinical trial were published in the May 16th issue of Science Translational Medicine.
“We are excited to have supported this clinical project with such promising results,” said Dr. Richard Snyder, Director of Florida Biologix and co-author on the publication. “Our specialized experience in the manufacture, testing and filling of biopharmaceuticals combined with our unique business model made us an ideal partner to work with the National Taiwan University Hospital. As with all of our clients, we worked closely with their team to achieve their manufacturing objectives.”
About Florida Biologix
Florida Biologix® (www.floridabiologix.ufl.com) is a Phase I/II biologics contract development, manufacturing and testing organization, based at the University of Florida’s Center of Excellence for Regenerative Health Biotechnology, that offers a range of HYPERLINK "http://www.floridabiologix.ufl.com/services/" biopharmaceutical services to the biotechnology industry and research institutes. They provide exceptional customer service, close project collaboration, and clear and honest communication. Florida Biologix’ in-house quality departments reside within the 23,000 square foot, state-of-the-art GMP facility, designed for multi-product bulk substance manufacturing, cell therapy product manufacturing and aseptic vial filling; an adjacent building houses the 5,000 square foot process development laboratories.
Florida Biologix staff are not only experts in biologics manufacturing, based on a foundation of science and engineering, but are also approachable, trustworthy, and believe that the best way to build long-term relationships with clients is through open communication and on-time delivery.