Role of the Institute
The Goldyne Savad Institute of Gene Therapy has successfully recruited topnotch scientists with high scientific achievements, each of whom is afforded full scientific independence and is responsible for the development of her/his research. Cross-fertilization of ideas among these scientists not only at the Institute but with scientists from other institutions and disciplines has culminated into important scientific activities.
Research
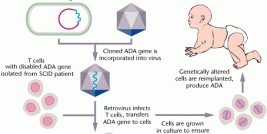
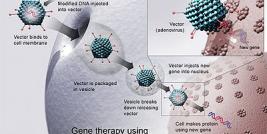
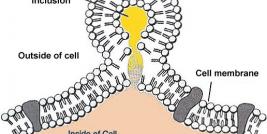
Research at the Institute covers a wide range of diseases along three main tracks: cellular gene therapy, viral vectors and non-viral vectors. In addition, the Institute is engaged in the development of research and biotechnological tools for gene therapy, which we hope will facilitate the use of gene therapy in the clinical setting. A substantial proportion of research at the Institute is done in cooperation with researchers from the Hebrew University, Weizmann Institute of Science and with researchers from other research centers in Israel and worldwide. Thus, the Institute provides the environment and activities necessary to facilitate cross-fertilization of ideas through the creation of an interdisciplinary community of researchers supporting one-another in the conception, initiation and development of research projects.
Translation
The mission of the Institute, as recommended by the Scientific Steering Committee and adopted by the Director of the Hadassah University Hospital, is to translate the fruits of laboratory research into therapies that will directly benefit patients. Hadassah's research laboratories are an inseparable part of the hospital's clinical department. The Institute is strategically placed to transfer new knowledge from the laboratory to the patient and back to the laboratory.
Education
The Institute seeks to train a new generation of scientists to explore the common elements of genetic-based disorders and to extend the concepts of basic genetics to their diagnosis and management. The aim of the Institute is not only to provide education to future scientists, but we expect our graduates to play leadership roles in the field of gene therapy. The Institute provides education and teaching to students pursuing M.Sc. or Ph.D. studies. For example, the Institute is a major player in the research training of undergraduates, graduate students, medical students and post-doctoral fellows. Presently, the Institute has 28 students (graduates and postgraduates) working with the PIs on different research projects. Because of the overlap in research interests among faculty, the trainees have the opportunity to interact with each other and with preceptors within a program representing a wide range of interests in gene therapy, from the most basic to the applied sciences, enhancing the breadth and depth of their training experience.
Guidelines
In an effort to expedite clinical application of gene therapy, the Institute has adopted a number of simple general guidelines:
- Recruiting Principal Investigators (PI's) with high scientific achievements, each of whom is afforded full scientific independence, and is responsible for the development of her/his research group;
- Focusing on areas in which the PIs have proven experience and expertise;
- Targeting and solving specific gene therapy bottlenecks by developing new technologies
- Conducting a tight collaborative effort between the groups.
In order to afford cross-fertilization of ideas on an international level, The Israeli Society of Gene Therapy was established on May 2002 as a medical research society which promotes studies in the field of gene therapy. The ISGT conducts annual meetings in the field of gene therapy. For more information click www.isgt.org.il.
Structure
The institute comprises three independent units, each focusing on a different stage in the development of a given gene therapy project:
The research laboratories currently accommodate 10 independent research groups. The laboratories conduct in vitro and in vivo studies, including the development of animal models and assessment of platform technologies.
The National-Hadassah gene therapy production facility. Once a project achieves reasonable maturity with a proven potential in an animal model it moves to the production step at the National-Hadassah Gene Therapy Production Facility. This facility is able to produce molecular therapeutics, including proteins, DNA, viral vectors and cell therapeutics, all meeting the current good manufacturing practice (cGMP) guidelines of the EMA and FDA. The facility is able to produce three lines of biological therapeutics simultaneously. Additional projects awaiting the production stage include the generation cGMP human embryonic stem cells for future research and potential clinical use, and a multiple sclerosis cell based vaccine project. The products will later be assessed in toxicological studies and applied in phase I clinical studies.
Clinical Research Ward
When the production process is completed, and the investigator's new drug file is approved, the product enters the clinical stage. The clinical Research Ward is currently conducting a number of Clinical studies.. Some phase I studies are part of research projects conducted by investigators at the institute. The facility also supports other research groups both at the Hadassah medical center and elsewhere.