Ark Therapeutics Group plc ("Ark" or the "Company") (AKT:LSE) today announces that the first Named Patient Supply (NPS) for Cerepro® has been approved by the French Medicines Control Agency (AFSSAPS) following a 'nominative' ATU application (Autorisation Temporaire d'Utilisation) made by a neuro-surgeon in France. Cerepro® (sitimagene ceradenovec), Ark's novel gene-based medicine, is being developed as an Orphan Drug for the treatment of operable malignant glioma.
Named Patient Supply through the ATU process in France is granted on an exceptional and temporary basis, for the use of a medicinal product without a marketing authorisation in the treatment, prevention or diagnosis of serious or rare diseases where no suitable therapeutic alternative exists and when the benefit/risk ratio of the medicinal product is presumed positive.
An earlier application to make Cerepro® available through the ATU process was denied in France in 2007. Since then Ark has completed Phase III development and has also filed a Marketing Authorisation Application (MAA) with the European Healthcare and Medicines Evaluation Agency (EMEA).
The Phase III study of Cerepro®, which completed in July 2008, showed that treatment with Cerepro® resulted in a significant therapeutic benefit, supporting the results of the previous Phase II clinical studies.
The MAA for Cerepro® was filed by Ark in late 2008 and the Company recently announced it had cleared the validation stage. The MAA is now undergoing formal review via the centralised procedure which is the standard route for all advanced therapies.
Dr David Eckland, R&D Director of Ark commented: "Cerepro® consistently demonstrates clinical benefits in trials and represents a much needed treatment for operable glioma. Ark is now receiving an increasing number of requests to treat patients with Cerepro® and we are delighted that this first approval has been given in France. The decision to approve on a Named Patient Supply demonstrates the increasing recognition of the utility of the product as well as the confidence amongst the neuro-surgeon community to use it."
Dr Nigel Parker, CEO of Ark, said: "We are very pleased with this initiative. There remains a significant unmet need in patients with malignant glioma and approval on a named patient basis is a logical step to making the product more widely available. This is an important milestone and another first for Ark as we believe it is the first time a gene medicine has been approved for use in a Named Patient Supply programme in Europe."
1. From: Notice to Applicants for Temporary Authorisation for Use, November 2007, AFSSAPS (website http://www.afssaps.sante.fr)
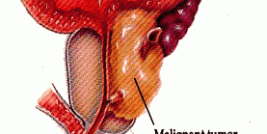
Malignant glioma
Malignant glioma is a devastating and fatal form of brain tumour that is usually confined to the brain. The current standard therapy involves surgically removing the solid tumour mass (when possible) and initiating radiotherapy and/or chemotherapy. Even with the latest approved treatments, most patients die within one year of diagnosis, with average survival being about eight months. Little therapeutic progress has been made in recent years and the prognosis for malignant glioma patients is poor. A high unmet clinical need exists for new treatments that prolong life in this devastating disease. There are approximately 16,000 cases of malignant glioma in the EU which are operable.
Cerepro®
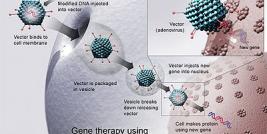
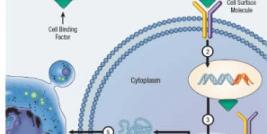
Cerepro® is an adenoviral mediated gene-based medicine (ad.HSV tk) given by multiple injections into the healthy brain tissue of patients following surgical removal of the solid tumour mass. In the following days, ganciclovir, is given intravenously. Once treated, healthy brain cells surrounding the site where the tumour was removed express the enzyme thymidine kinase. This converts the ganciclovir to a substance which specifically kills dividing cells. The healthy neurones surrounding the tumour in the brain are non-dividing and are therefore not susceptible to this substance. In this way Cerepro® harnesses healthy brain cells to help prevent a new tumour from growing.
Ark Therapeutics Group plc
Ark Therapeutics Group plc is a specialist healthcare group (the "Group") addressing high value areas of unmet medical need within vascular disease, wound care and cancer. These are large and growing markets, where opportunities exist for effective new products to generate significant revenues. With five marketed devices, Kerraboot®, Kerraped®, Flaminal®, Kerramax® and Neuropad®, and three further lead pharmaceutical products in late stage clinical development: Cerepro®, Vitor™, and Trinam®, the Group is transitioning from an R&D company to a commercial, revenue generating business.
Ark's own products are sourced from related but largely non-dependent technologies within the Group and have been selected to enable them to be taken through development within the Group's own means and to benefit from Orphan Drug Status and/or Fast Track Designation, as appropriate. This strategy has allowed the Group to retain greater value and greater control of clinical development timelines, and to mitigate the risks of dependency on any one particular programme or development partner. Ark has secured patents or has patent applications pending for all its lead products in principal pharmaceutical markets.
Ark has its origins in businesses established in the mid-1990s by Professor John Martin and Mr Stephen Barker of University College London and Professor Seppo Yla-Herttuala of the AI Virtanen Institute at the University of Kuopio, Finland, all of whom play leading roles in the Company's research and development programmes.
Ark's shares were first listed on the London Stock Exchange in March 2004 (AKT.L).
This announcement includes "forward-looking statements" which include all statements other than statements of historical facts, including, without limitation, those regarding the Group's financial position, business strategy, plans and objectives of management for future operations (including development plans and objectives relating to the Group's products and services), and any statements preceded by, followed by or that include forward-looking terminology such as the words "targets", "believes", "estimates", "expects", "aims", "intends", "will", "can", "may", "anticipates", "would", "should", "could" or similar expressions or the negative thereof. Such forward-looking statements involve known and unknown risks, uncertainties and other important factors beyond the Group's control that could cause the actual results, performance or achievements of the Group to be materially different from future results, performance or achievements expressed or implied by such forward-looking statements. Such forward-looking statements are based on numerous assumptions regarding the Group's present and future business strategies and the environment in which the Group will operate in the future. Among the important factors that could cause the Group's actual results, performance or achievements to differ materially from those in forward-looking statements include those relating to Ark's funding requirements, regulatory approvals, clinical trials, reliance on third parties, intellectual property, key personnel and other factors. These forward-looking statements speak only as at the date of this announcement. The Group expressly disclaims any obligation or undertaking to disseminate any updates or revisions to any forward-looking statements contained in this announcement to reflect any change in the Group's expectations with regard thereto or any change in events, conditions or circumstances on which any such statements are based. As a result of these factors, readers are cautioned not to rely on any forward-looking statement.